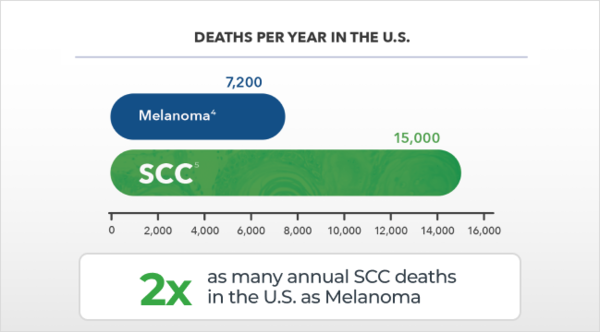
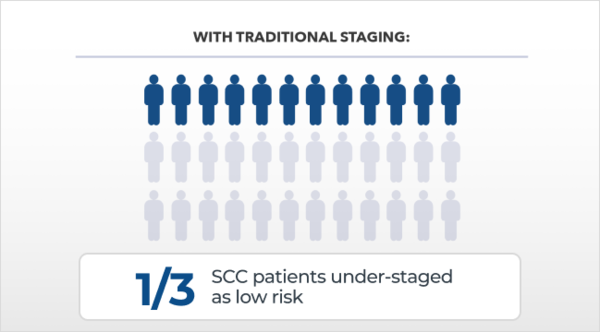
Predicting risk of metastasis for SCC is not precise, DecisionDx-SCC is.
A complement to current staging methods, DecisionDx-SCC is the strongest independent predictor of cutaneous squamous cell carcinoma (SCC) metastasis.1
A diagnosis is clear-cut. Charting next steps can be more challenging. Because cancer treatment, management plans and their outcomes are guided by risk of metastasis, prognostic accuracy has direct implications on patient management and outcomes.
Whether you’re determining referrals, treatment, or frequency of follow-up care, it’s imperative to have the most valuable prognostic data available.

Accurate prognostic data you need
DecisionDx-SCC is a Gene Expression Profile (GEP) test developed to predict nodal and distant metastatic risk for SCC patients with one or more high-risk factors, based on the biologic profile of 40 genes within their tumor tissue.
DecisionDx-SCC looks at the biological behavior of a patient’s tumor utilizing a neural network algorithm to classify patients into three molecular risk profiles.2 The test, which can be used independently or to complement traditional staging, provides additional risk prediction to better inform choices about treatment and follow-up care.
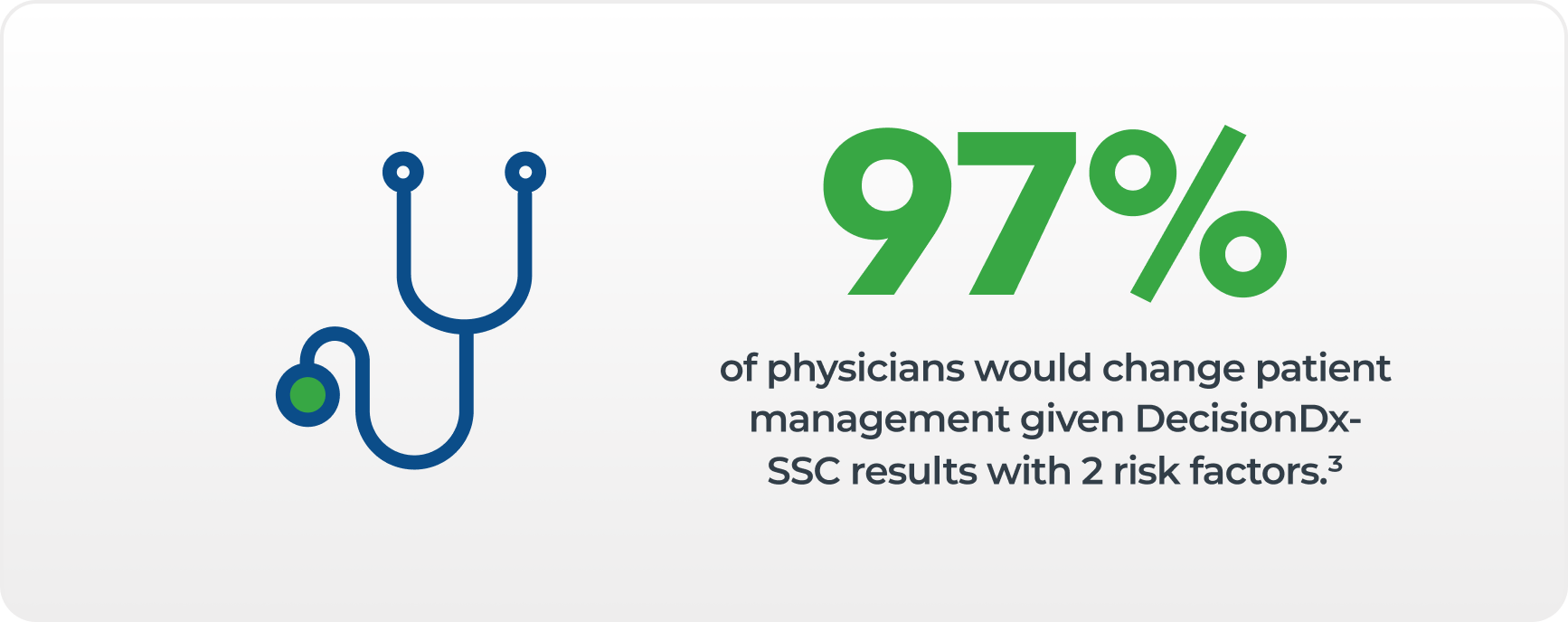

DecisionDx-SCC is clinically validated in nearly 1,000 patients in two independent validation studies, and supported by 17+ peer-reviewed publications.1,7
More than 3,500 clinicians have ordered DecisionDx-SCC*
Intended Use: DecisionDx-SCC is indicated for patients diagnosed with cutaneous squamous cell carcinoma (SCC) and one or more risk factors.8,9 The personalized class results complement current staging methods commonly used for risk assessment.
*Castle Biosciences data on file


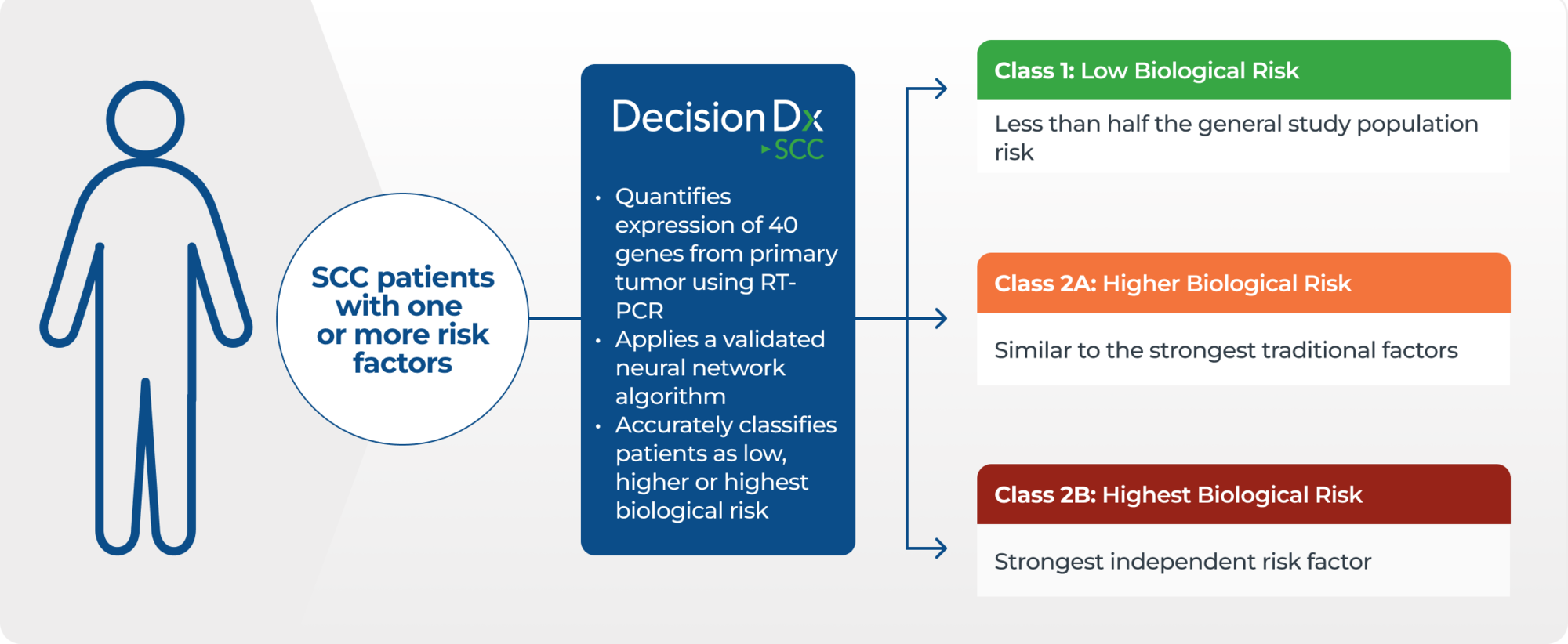
The test provides an accurate and comprehensive result with higher positive predictive value (PPV) than traditional staging, adding valuable prognostic information that is multiplicative to existing risk seen in traditional clinicopathologic risk factors.1
You can order DecisionDx-SCC in any SCC patient with one or more of the following risk factors.

Risk Factors:
Surgical and Pathological Findings
-
Perineural involvement:
- Large ( ≥ 0.1 mm) or named nerve involvement
- Small (≤ 0.1 mm) in caliber
- Poorly differentiated tumor histology
-
Depth:
- Invasion beyond subcutaneous fat
- Invasion depth ≥2 mm
- Clark level ≥IV
- Aggressive histologic subtypea
- Lymphovascular invasion
- Desmoplastic SCC
- DecisionDx-SCC is not intended for use with locally recurrent tumor tissue.a) Acantholytic (adenoid), adenosquamous (showing mucin production), or carcinosarcomatous (metaplastic) subtypes8 (others9 will be considered on a case-by-case basis)
History and physical examination
- Tumor size ≥2 cm anywhere on the body
- Tumor location on the head, neck, hands, genitals, feet or pretibial surface (areas H or M)
- Immunosuppression
- Rapidly growing tumor
- Tumor with poorly defined borders
- Tumor at site of prior radiation therapy or chronic inflammation
- Neurologic symptoms in region of tumor

Case study:
Two real-world SCC patients had similar risk profiles but very different outcomes. When applying the DecisionDx-SCC gene expression profile test, the limitations of traditional risk factors and staging methods become apparent.10
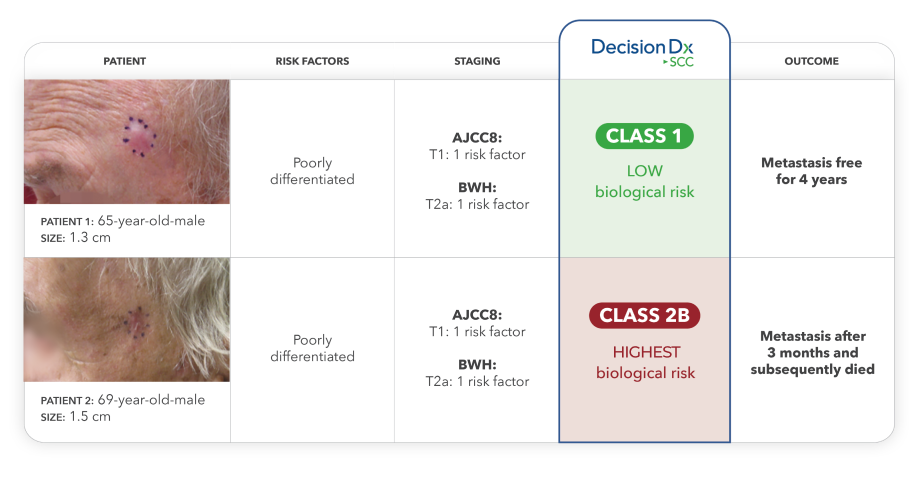
Understaging poses challenges for clinicians and their patients
35% of cases with metastasis are under-called by staging and can lead to an unforeseen metastasis in traditionally low-risk SCC patients.6
DecisionDx-SCC can be used independently or alongside traditional staging to help dermatologists, NPs and PAs:
- Ensure patient's personalized risk of metastasis is appropriately identified so they can receive the right treatment at the right time
- Deliver personalized treatment to each patient based on their underlying tumor biology
- Provide more information and treatment options that match patient’s individualized demographics
- Avoid unnecessary treatment interventions on lower-risk SCC patients
- Contribute to patients’ and systems’ healthcare savings
What are my next steps?
Learn more about ordering a test.

Inform decisions,
improve care.
Learn more about integrating DecisionDx-SCC into your practice
Find out more about Castle Biosciences
Talk to your doctor to determine if the DecisionDx-SCC test is right for you.
1. Ibrahim et al. Future Oncology 2021.
2. Singh et al. Clinical, Cosmetic and Investigational Dermatology 2023.
3. Goldberg et al. Presented at Winter Clinical Dermatology. January 14-19, 2022.
4. SEER Data Release 2019.
5. Mansouri B et al. JAMA Dermatology 2017.
6. Farberg et al. Dermatology and Therapy 2022.
7. Wysong et al. Dermatology and Therapy 2024.
8. NCCN Guidelines for Squamous Cell Skin Cancer, v1. 2024.
9. Connolly SM et al. JAAD 2020.
10. Au et al. Dermatology and Therapy 2021.
Copyright ©2024 Castle Biosciences